
Aseptic Preparation
Components after washing should be handled in at least a grade D environment. Handling of sterile starting materials and components, unless subjected to sterilisation or filtration through a micro-organism-retaining filter later in the process, should be done in a grade A environment with grade B background. ...

Line Clearance Checklist for Secondary Packaging Area
1 Ensure that product containers, materials, status labels, BPR of previous process operation are removed.
2 Ensure that area is cleaned as per the cleaning procedure and records are updated.
3 Ensure that temperature, relative humidity and differential pressure of the area is suitable for dispensing.
4 Ensure ...

Type of Tablet Press
There are Manual and Automatic Tablet Press
Manual Tablet Press : Generally, is of single punch or multi-tip single punch. They are mostly used for research and/or initial pharmaceutical product development work for evaluating API’s compaction characteristics for direct compression or dry granulation ...

Procedures for Pre-entering Aseptic Area
Sterile products administered by injection or infusion inorder to bypass the gastrointestinal tract. Administration involves the use of a needle topenetrate the skin. Parenteral administration technically means non-oral administration, but ittypically refers to the injection of drugs into the body intravenously, ...

Access to the Aseptic area
Only authorized specially trained personnel are permitted to enter thesterile and aseptic manufacturing area. Any condition which may cause the shedding ofabnormal numbers or types of organisms (e.g. coughs, colds or any other type of infection) mustbe reported by the employee to the supervisor who will ...

Aseptic Sampling and Dispensing of Sterile Materials
Aseptic Sampling & Dispensing Area: The aseptic sampling and dispensing of raw materialsmust be carried out in the Laminar down Flow / Cross Flow Unit (LAF cabinet / Down FlowHood) within the dispensing room. Access to the aseptic area is permitted only via thepersonnel air lock. Only one batch ...

Pharmaceutical Liquid Preparations
Liquid dosage forms are designed to provide the maximum therapeutic response in a target populationwith difficulty swallowing tablets and capsules and/or to produce rapid therapeutic effects.Liquid dosage forms are either, internal, parental or external use. They are available in monophasic andbiphasic ...
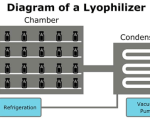
Lyophilization Cycle
• Freezing – Filled vials are loaded at 5°C (shelf temperature) and the product freeze by reducingthe shelf temperature to -45°C (at 0.2°C/min). Then hold at -45 °C for a duration of not less than240 minutes.
• Annealing – After freezing, the product is subjected to heat annealing by ...

Lyophilization Process
Lyophilization or freeze drying is a process in which water is removed from a product after it is frozenand placed under a vacuum, allowing the ice to change directly from solid to vapor without passingthrough a liquid phase. The process consists of three separate, unique, and interdependent processes;freezing, ...

Sterilisation by Radiation
Radiation sterilisation is used mainly for the sterilisation of heat sensitive materials and products. Many medicinal products and some packaging materials are radiation-sensitive, so this method is permissible only when the absence of deleterious effects on the product has been confirmed experimentally. ...

