
Stability analysis
Stability analysis is an area of great interest in preformulation studies. It is a complex set of procedures performed on candidate drug molecules or dosage forms for their final immediate packaging intended for marketing and, where appropriate, novel formulation excipients. By investigating the ...

Sample Testing Procedure
Manager QC or his designee shall assign the analytical work and allot samples to trained and approved analyst.
After getting the sample allotted, the analyst shall collect analytical worksheet/s depending on the number of samples, and get the same approved by QC manager or designee.
The analyst shall ...

Long-term stability studies
Long-term, or real-time, stability testing programs subject products to recommended storage conditions and typically use a longer observation period for degradation to establish the product’s shelf-life. This testing method differs from accelerated shelf-life testing, which instead exposes products ...

In-Process Quality Control (IPQC)
In-process quality control tests are simply routine tests that are performed during production. They are the tests that are carried out before the manufacturing process is completed to ensure that the products meet the established product quality before they are approved for use and marketing.
The function ...

10 Tips for HPLC Analysis In Pharmaceuticals
HPLC, also known as High-Performance Liquid Chromatography system, is one of the most crucial analytical methods in pharmaceuticals. The purpose of HPLC is the separation of each compound that make up a mixture.
It passes pressurized liquid solvent with the use of pumps which holds the sample mixture ...

Quality control area
1 The quality control laboratory should be separated from the production area. Where biological, microbiological or radioisotope test methods are applied they should be separated from each other.
2 Quality control laboratories should be designed to suit the operations carried out in them Sufficient space ...

Difference Between QC and QA
Unlike quality control, quality assurance is usually focused on how processes are performed or how services are delivered. It focuses on preventing mistakes made by creating and evaluating processes, strategies and brand policies. Typically, your quality assurance function will have a “checklist” ...
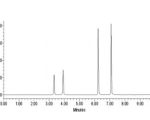
System Suitability Test (SST)
Selecting a proper System Suitability Testing mixture is essential to check the specifications of a liquid chromatographic system. System Suitability Testing limits are acceptance criteria that must be met prior to sample analysis. Some of the common chromatographic systems used in pharmaceutical sample ...
Particle Counter
A particle counter is used to monitor and diagnose particulate contamination in certain cleaning media, including air, water, and chemicals. Particle counters are used in a variety of applications to support clean manufacturing practices, industries including: electronic components and assemblies, pharmaceutical ...

Apparatus of HPLC
“Basic overview of HPLC process” and its procedures are now covered In more detail, HPLC consists of various components including a solvent delivery pump, a degassing unit, a sample injector, a column oven, a detector. , and a data processor. HPLC flow diagram showing the role of each component.
As ...

